44 grams = 0.0970024 pounds. 1 mole of propane is 44 grams and 1 mole of water is 18 grams.

Flame King 20 Lb (Pound) Steel Buffer Cylinder With Fill And Vapor Valve - Walmart.com
In imperial or us customary measurement system, the density is equal to 30.777 pound per cubic foot [lb/ft³], or.
How much water vapor produced in 1 lbs of propane. Assuming that all volume measurements occur at the same temperature and pressure, how many liters of oxygen will be required to completely burn. The molar mass of propane is about 44 grams/mol. 30.77698 pounds [lbs] of propane fit into 1 cubic foot.
Using propane for the most efficient water heaters on the market. 7.72 g h_2o the nie for the complete combustion of propane is: The water capacity is how much water the propane tank will hold in pounds.
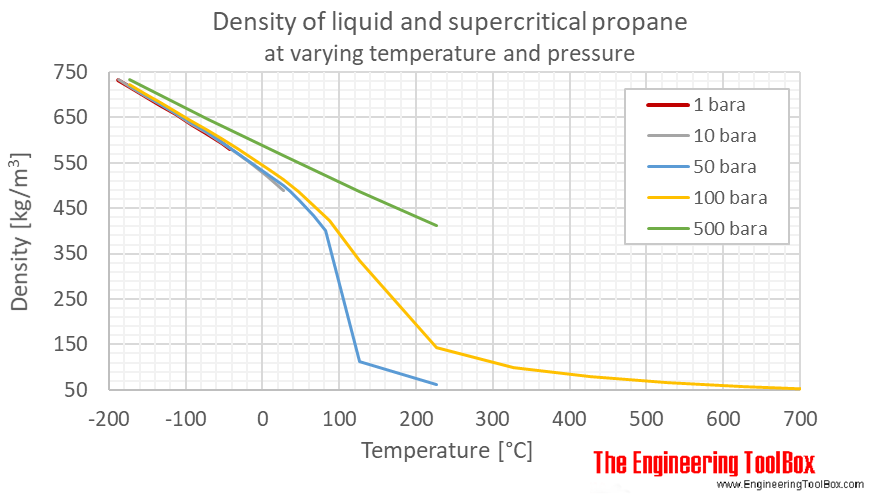
For liquid propane, it varies according to temperature. 0.09070024 / 0.1587312 = 1/x. 10000/4 *0.70 = 1750 btu.
The pistons turn the drive shaft, and the drive shaft turns the wheels. For example, the “wc” stamped on the tank followed by a number such as “47.6” means the tank will hold 47.6 pounds of water. Vaporization is affected and influenced by the actual size of the propane tank.
Average 70% on a 15 min roast. To find out how much that is, we need to convert liters of o_2 to moles of o_2 using the fact that at stp, a mole of gas is. Propane works the same way but at a much lower boiling point.
4.56 gal x 4.24 lbs/gal propane is equal to 20 lbs of propane. 950 grams of water / 13.7 = 69 grams of water. The tank will hold 20 lbs of propane and maintain 20% head space.
Lpg density or specific gravity is about half that of water, at 0.51. Water vapor in air ( pounds water vapor per pound dry air) relative humidity. At 77 degf, it is 1 gallon per 4.11#.
1 kg gas roaster, 10.000 btu max. A gallon of propane gives about 90,000 btu, burning a gallon of propane produces about a gallon of water (in vapor). 18 grams = 0.0396828 pounds x 4 = 0.158731 pounds.
Vapour (or vapor in american spelling) is the more technically correct term for lpg, as it is. 1 liter of propane = 24000 btu. 1 gallon of gasoline weights 8.66lbs, of which 1.4 lbs is hydrogen, as gasoline contains 16% hydrogen and.
14 votes) moisture buildup can be a big problem in tiny spaces during the winter. Propane cylinder (approx) pounds of propane in cylindermaximum continuous draw in btu/hr at various temperatures in degrees f. * grains of water is commonly used in psychrometrics.
Propane weighs 0.493 gram per cubic centimeter or 493 kilogram per cubic meter, i.e. One might refer to propane vapor as lp gas steam. Propane gas (c 3 h 8) burns completely in the presence of oxygen gas (o2) to yield carbon dioxide gas (co 2) and water vapor (h 2 o).
X= 1.75 pounds of water for each pound of propane. Dividing 4.24 by 8.33 equals.51. In australia, where lpg is propane, 1kg of lpg has a volume of 1.96l.
Burning 1 gallon of gasoline produces 1.52 gallons of water. Write a balanced equation for this reaction. C 3 h 8 (propane) + 5o 2 (oxygen) → 3co 2 (carbon dioxide) 4h 2 o (water) for each molecule of propane burned on the left, 4 molecules of water are released on the right.
There are 226.77 centimoles in 100 grams of propane. In a gasoline engine, a mixture of gasoline and air is combusted to drive pistons. Molecular weight of oxygen=32 so 44 lb of propane will require (32 x5)= 160 lb of oxygen for complete combustion to water and co2.
2 3 professionally accredited training the propane education & research council (perc). At 25°c (77°f or 298.15k) at standard atmospheric pressure. 4) after the above mentioned procedures you will be able to know the pressure at any container temperature as well as the amount of produced vapor at such pressure when all.
One gallon of water weighs 8.33 lbs. Propane is heavier than air and will seek the lowest space available. Density of propane is equal to 493 kg/m³;
At normal pressure it liquifies below its boiling point at −42 °c and solidifies below its melting point at −187.7 °c. Propane weighs 0.493 gram per cubic centimeter or 493 kilogram per cubic meter, i.e. If you are venting to the outside of the house, as soon as the vent gets hot and a good draw is established then you should not have to worry about the water vapor in the house.
Water boils at 212°f at which point it converts to a vapor we know as steam. If instead you intended methane in the liquid state (but it's a very unusual condition!) then from 1 gallon of methane you have 0.932 gallons of liquid water. 0° 20° 40° 60° 70° 100 113,000 167,000 214,000 277,000 300,000 90 104,000 152,000 200,000 247,000 277,000
Molecular weight of propane=44 2. • condensing tankless water heaters: Propane crystallizes in the space group p2 1 /n.
For water, 1 gallon = 8.33#. What observable change would indicate that a chemical change occurred? I need help with this problem:
Unlike water, 1 kilogram of lpg does not equal 1 litre of liquid lpg. Therefore 20 lb of propane will require= 20/44 x. Conversely, 1l of lpg weighs 0.51kg.
Density of propane is equal to 493 kg/m³; During a normal roast the combustion of propane gives off 69 grams of water. This means propane is less dense than water (lighter than water) and.
493 kilograms [kg] of propane fit into 1 cubic meter. As propane boils, it is in the process of vaporizing. The low spacefilling of 58.5% (at 90 k), due to the bad stacking properties of the molecule, is the reason for the particularly low melting point.
1 grain = 0.000143 lbm = 0.0648 g (7000 grains per lbm) same table as above with water vapor in pounds water vapor per pound dry air. $17~\mathrm{l}$ of propane are incompletely combusted in $34~\mathrm{l}$ of oxygen at $25~^\circ\mathrm{c}$ and $100~\mathrm{kpa}$ into only water and carbon. 24000 btu / 1750 btu = 13.7.
What is the mass of the water produced? Let's take a look at the chemistry. Propane is a colorless, odorless gas.
Write a balanced chemical question to represent this including state symbols. In the combustion reaction, gasoline (aka, octane, c8h18) combines with atmospheric oxygen (o2) to produce carbon dioxide (co2) and water (h20): At 25°c (77°f or 298.15k) at standard.

Lp Or Natural Gas Fired Appliance Combustion Products Found In Flue Gas Exhaust

Propane Reacts With Oxygen Gas To Produce Water Vapor And Ca By Shairah Segne

Flame King 5 Lbs. Empty Propane Cylinder With Overfill Protection Device Ysn5Lb

How Much Water Does Burning Propane Produce? - Tiny Wood Stove

Propane - Density And Specific Weight Vs. Temperature And Pressure