One would expect that for low temperatures, temperature vs. The water cooled to a temperature of 14°c in 30 minutes.
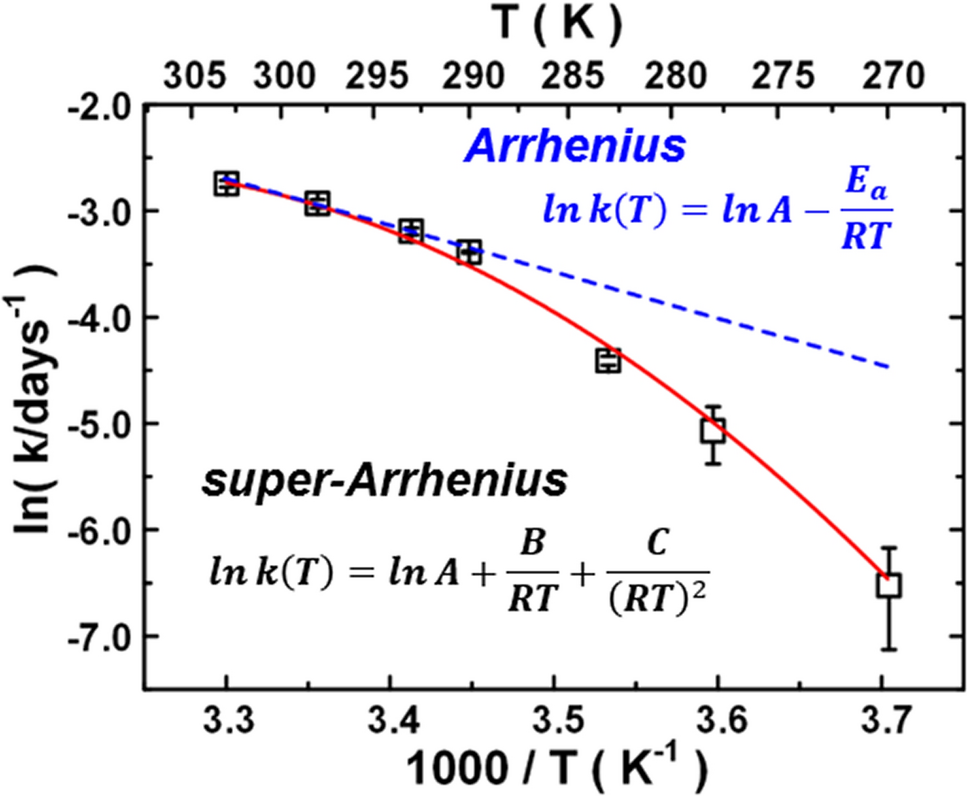
Temperature Effect On Water Dynamics In Tetramer Phosphofructokinase Matrix And The Super-Arrhenius Respiration Rate | Scientific Reports
Thermal expansion is the tendency of matter to change its shape, area, volume, and density in response to a change in temperature, usually not including phase transitions.

Water heating temperature exponentia. If the water was losing heat in proportion to how much hotter it was than some cold sink, and at the same time you added. Calculating the log mean temperature difference, lmtd (∆t lm)fig. Look for any signs of deformity in the top or bottom surfaces of the water heater, or in the relief line or water connection.
A practical application is that it can tell us how fast a water heater cools down if you turn off the breaker when you go on vacation. When a substance is heated, molecules begin to vibrate and move more, usually creating more distance between. Then by newton’s law of.
As far as temperature is concerned, this is far more complex. We waved the thermometer so that the air aorund it was not heating up. 30 gallons + 0.351 gallon = 30.351 gallons.
It depends on the setup. For editing and accuracy comments, june 2012. 220.6 bar = 22.06 mpa.
Ensure the thermometer is about 2cm above the bottom of the beaker. 488 kwh for 1 hour. The overall heat transfer coefficient is 155 w/m2.k and the exchanger area is 10m2.
If the water were totally insulated and you added heat at a constant rate, then the temperature would increase linearly with time. The enthalpy of vaporization is a function of the pressure at which that transformation takes place. For a microwave rated 1,300 watts and a typical 50% efficiency$^1$ that means a constant 650 watts of heat energy delivered to the water.
Online water specific heat calculator. Many water heaters are set to 140 degrees per. 21.2.1 a cup of water was heated to a temperature of 70°c and placed in a refrigerator that had a temperature of 8°c.
Energy e = m•cp•δt = 7000 x 4.186 x 60 = 1758120 kj. If the water exits at 44°c, determine the exit temperature of the helium and its mass flow rate. $\delta t= \frac{q}{mc}$ the heat.
The thermophysical properties of the base fluid water and. To heat the same water volume in half the time (30 minutes) would need twice the heating power, ie, 7kw. The perfect temperature to set a hot water heater.
I did an exponential regression on the data (well, appropriately shifted data). On the other hand, the epa suggests turning the thermostat down to 120 to reduce power consumption, and a booster heater to reach sanitizing temperatures at select outlets. Time graph to be linear.
To calculate the change in temperature $(\delta t)$ of a mass of material $(m)$ based on how much heat energy $(q)$ was added, you use the material's heat capacity $(c)$: 30 us gallons of oil is heated from 7 0of to 100of. Imagine i am trying to heat water from room temperature to 100 degrees celsius, with a heating element of constant power.
This is a nonlinear process as the vapour pressure of water is nonlinear with temperature and the rate of evaporation is linear with. Energy required for heating the water to 120°f: When the temperature on the thermometer has reached 90°c, immediately switch off the burner.
So, the energy required to raise the temperature of 7000 kg of water from 20c to 80c is: When water heater thermal expansion creates dangerously high water pressure, it can cause the top or bottom of the tank to bulge or rupture. 373.946 °c or 705.103 °f, critical pressure:
Microwaves are absorbed in the outer layer of foods, and so still rely on conduction to heat internally. Light the bunsen burner and put on a blue flame. When i did the experiment, the result however was linear up to about 60 degrees, but then it started to look more exponential.
Converesely, if we only use half the heating power, 1.75kw, it will take twice as long to heat up to desired temperature, ie, 2 hours. A distorted water heater is quite dangerous and needs to be replaced. = 100 gal day × 8.3 lb gal ︸ m × 1 btu lb °f ︸ c p ×.
If we only have a 1kw element available, we will expect a heat up time in excess of 3 hours. Suppose that a body with initial temperature t 1 °c, is allowed to cool in air which is maintained at a constant temperature t 2 °c. After 60 minutes the water will be about 5.6°c.
Move the cursor beside l and press [solve]. Water heaters are factory set at osha recommended levels, around 140 degrees. In mathematics, if given an open subset u of r n and a subinterval i of r, one says that a function u :
Heat required (btu) = m x c p x (temperature difference) where c p is the heat capacity of water (1 btu/lb/f) and m is the mass of the water (assume 1 gal has 8.3 lb of water and the 3,412 btu = 1 kwh) solution: The heat of vaporization diminishes with increasing temperature and it vanishes completely at a certain point called the critical temperature (critical temperature for water: In this case, since we are talking of heat transfer, it's the newton's law of cooling, but equations of the same form are very common.
Power = energy / time. Let the temperature of the body be t°c at time t. Temperature is a monotonic function of the average molecular kinetic energy of a substance.
If you're looking for a short answer, it's 120 degrees fahrenheit (about 49 degrees celsius). This is just a classical exponential decay and obeys to a very precise low. Hot water at 80°c enters the tube of two shell pass, eight tubes pass h.e at the rate 0.375 kg/s heating helium from 20°c.
Find the temperature of the water after 60 minutes. For a microwave the transfer of energy is linear if there is something in the chamber to absorb the microwaves. Temperature t as a function of time t of a thermometer initially at temperature t0 immerged in a liquid at temperature tc at time t = 0.
U × i → r is a solution of the heat equation if = + +, where (x 1,., x n, t) denotes a general point of the domain. And this is 488.36 kilo watts of power (since 1j/s = 1w) 1758120 kilojoule/hour = 488.36666667 kilowatt hour. Thanks to careful reader anthony lee in the u.k.
= m × c p × δt. From another point of view, lower temperatures are better for. Water will also quickly heat from freezing to boiling point, then.
The calculator below can be used to calculate the liquid water specific heat at constant volume or constant pressure and given temperatures. It is typical to refer to t as time and x 1,., x n as spatial variables, even in abstract contexts where these phrases fail to have.